Chapter 15: Equilibrium
- Equilibrium occurs when the forward and reverse reaction proceed at the same rate.
- Equilibrium doesn't mean the amount of reactants and products is equal, it means they're constant.
- When writing a system at equilibrium, it's equation must have double arrows.
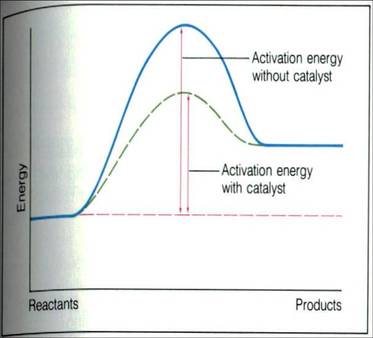 Catalyst increase the rate of both the forward and reverse reactions.
Catalyst increase the rate of both the forward and reverse reactions.
The Equilibrium Constant
Example 1: Using the ICE method (Initial, Change, Equilibrium)
A closed system initially containing 1.00 x 10^-3 M H2 and 2.000 x 10^-3 M I2 at 448C is allowed to reach equilibrium.
Analysis of the equilibrium mixture shows that the [HI] is 1.87 x 10^-3 M.
Calculate Kc at 448C for the reaction taking place :
H2(g) + I2(g) ⇌ 2HI(g)
I 1x10^-3 2x10^-3 0 *The starting amount is 0 because the reaction hasn't occured yet
C -9.35x10^-4 -9.35x10^-4 +1.87x10^-3 *1.87x10^-3 M HI x (1 H2/ 2 HI) = 9.35x10^-4 You have to divide by 2 because its a
E 6.5x10^-5 1.07x10^-3 1.87x10^-3 1:2 mole ratio between the reactants and product
Kc= [HI]^2 / [H2]x[I2] = (1.87x10^-3)^2/ (6.5x10^-5) x (1.07x10^-3) = (3.49x10^-6)/(6.96x10^-8)= 50.3 *K doesn't have any units
Product favored because K>1
- aA+bB ⇌ cC+ dD would become Kc= ([C]^c [D]^d)/([A]^a [B]^b]).
- Kc is just the products/reactants
- You only pay attention to aqueous and gas solutions. NOT solids or liquids.
- K>>1 reaction is product favored; product predominates at eq
- K<<1 reaction is reactant favored; reactant predominates at eq
- K=1 products and reactants are equal
Example 1: Using the ICE method (Initial, Change, Equilibrium)
- Make a table and fill in givens
- Fill in assumptions
- Use coefficients and stoichiometry
- Add data
- Complete
- Calculate Kc
A closed system initially containing 1.00 x 10^-3 M H2 and 2.000 x 10^-3 M I2 at 448C is allowed to reach equilibrium.
Analysis of the equilibrium mixture shows that the [HI] is 1.87 x 10^-3 M.
Calculate Kc at 448C for the reaction taking place :
H2(g) + I2(g) ⇌ 2HI(g)
I 1x10^-3 2x10^-3 0 *The starting amount is 0 because the reaction hasn't occured yet
C -9.35x10^-4 -9.35x10^-4 +1.87x10^-3 *1.87x10^-3 M HI x (1 H2/ 2 HI) = 9.35x10^-4 You have to divide by 2 because its a
E 6.5x10^-5 1.07x10^-3 1.87x10^-3 1:2 mole ratio between the reactants and product
Kc= [HI]^2 / [H2]x[I2] = (1.87x10^-3)^2/ (6.5x10^-5) x (1.07x10^-3) = (3.49x10^-6)/(6.96x10^-8)= 50.3 *K doesn't have any units
Product favored because K>1
The Reaction Quotient
Example 2:
Say you have 2.00 mol/L of H2, 1.00 mol/L N2 and 2.00 mol/L NH3 for
N2 + 3H2 ⇌ 2NH3 Kc= .105 at 472C
Q= [NH3]^2 / [H2]^3 [N2] = (2.00)^2/ (2.00)^3 (1.00) = 0.500
Q: 0.500 > K: 0.105 the equilibrium will shift to the left
- Allows you to predict which direction the reaction must go to reach equilibrium.
- It's used to check that a reaction is at equilibrium
- If Q=K the system is at equilibrium
- If Q > K there is too much product, equilibrium shifts to the left
- If Q < K there is too much reactant, equilibrium shifts to the right
- To calculate Q you must substitute the initial concentration of reactants and products into the equilibrium expression.
Example 2:
Say you have 2.00 mol/L of H2, 1.00 mol/L N2 and 2.00 mol/L NH3 for
N2 + 3H2 ⇌ 2NH3 Kc= .105 at 472C
Q= [NH3]^2 / [H2]^3 [N2] = (2.00)^2/ (2.00)^3 (1.00) = 0.500
Q: 0.500 > K: 0.105 the equilibrium will shift to the left
Chapter 16: Acid-Base Equilibria
When Acid Dissolves in Water
Example 1.
HNO2(aq) + H2O ⇌ NO2-(aq) + H30+(aq)
- Water acts as a Bronsted-Lowry base and extracts a proton (H+) from the acid.
- This results in the formation of the conjugate base of the acid and a hydronium ion.
Example 1.
HNO2(aq) + H2O ⇌ NO2-(aq) + H30+(aq)
- Remove H+
- Add H+
- HNO2(aq) = Acid
- H2O (l) = Base
- NO2-(aq) = Conjugate Base
- H30+ (aq) = Conjugate Acid
|
Lewis Acids
|
Lewis Bases
|
Acid and Base Strength
- Strong acids completely split in water.
- The conjugate base of a weak acid has to be strong and the conjugate base of a strong acid has to be weak.
- Weak acids only split partially in water.
- Non-acidic substances do not split in water.
- In an acid-base reaction, equilibrium will favor the reaction that moves the proton to the stronger base.
- Ka values are used to compare the strengths of weak acids; the greater the K the greater the strength
pH
pH=-log[H+]
pOH=-log[OH-]
pKa=-log Ka
- p(anything)=-log(anything)
pH=-log[H+]
pOH=-log[OH-]
pKa=-log Ka
- As pH decreases, [H+] increases
- When [H+]=[OH-], the solution is neutral, pH=7
- When [H+] is >[OH-], the solution is acidic, pH less than 7
- When [H+] is < [OH-], the solution is basic, pH greater than 7
- Since pH is based on a log scale, every 1 unit change in pH is 10 times the concentration.
|
pH
Strong Acids
-HCl, HBr, HI, HNO3, H2SO4, HClO3, HClO4
HA ⇌H+ + A-
|
pOH
Weak Acids
|
Autoionization of Water
- Water is considered both an acid and a base
- Water autoionizes by donating a proton to another water molecule.
- Also called amphoteric or amphiprotic
- Amphoteric substance reacts as a base when combined with something more acidic than it
Chapter 17: Additional Aspects of Acid/ Base Equilibria
The Common Ion Effect
Example 1.
HC2H3O2 ⇌ H+ + C2H3O2-
If you add more C2H3O2- the H+ decreases causing a shift to the left, therefore increasing the reactant.
Example 2.
Calculate the pH of a solution containing 0.085M nitrous acid (HNO2; Ka=4.5x10^-4) and 0.10 M potassium nitrite, KNO2.
HNO2 ⇌ H+ + NO2
I 0.085 M 0 0.10 M
C -x +x +x
E 0.085 x 0.10
Ka= (products) / (reactants) = (x)(0.10)/(0.085)=.1x/.085
.1x/.085= 4.5x10^-4
.1x=3.825e-5
x=3.825e-4
pH=-log(x)=-log(3.825e-4)= 3.42
Buffered Solutions
Buffer Capacity and pH Range
Example 3.
If pKa = 6 then the Buffer Range= 5 thru 7
- "The extent of ionization of a weak electrolyte is decreased by adding to the solution a strong electrolyte that has an ion in common with the weak electrolyte."
Example 1.
HC2H3O2 ⇌ H+ + C2H3O2-
If you add more C2H3O2- the H+ decreases causing a shift to the left, therefore increasing the reactant.
- The weak electrolyte will split up less if you add more C2H3O2
Example 2.
Calculate the pH of a solution containing 0.085M nitrous acid (HNO2; Ka=4.5x10^-4) and 0.10 M potassium nitrite, KNO2.
HNO2 ⇌ H+ + NO2
I 0.085 M 0 0.10 M
C -x +x +x
E 0.085 x 0.10
Ka= (products) / (reactants) = (x)(0.10)/(0.085)=.1x/.085
.1x/.085= 4.5x10^-4
.1x=3.825e-5
x=3.825e-4
pH=-log(x)=-log(3.825e-4)= 3.42
Buffered Solutions
- Solutions with weak conjugate acid-base pairs can resist drastic changes in pH by adding small amounts of strong acid or base
- Resist pH changes because they contain an acidic species to neutralize OH- ions and a basic species to eliminate the H+ ions
- The acidic and basic species that make up the buffer do not neutralize each other
- Buffers are often made by mixing a weak acid or weak base with a salt of that acid or base
- By choosing the appropriate components and adjusting the concentrations, you can make a buffer solution at any pH
- The pH is determined by the Ka for the weak acid and the ratio of the concentrations for the conjugate acid-base pairs
- You can use the process from the common ion effect to calculate the pH
Buffer Capacity and pH Range
- These are the 2 important characteristics
- Buffer capacity depends on the amount of acid and base from which it is made
- The pH range of any buffer is the pH range over which the buffer acts effectively
- Buffers resist a change in pH in either direction when the concentration of the weak acid and conj. base are about the same.
- When concentration of weak acid = concentration of conj. base then pH=pKa
- Buffers usually have a range of +/- 1pH units of pKa
Example 3.
If pKa = 6 then the Buffer Range= 5 thru 7
Works Cited:
Floden, Mark. "Ch1011 Module 1." Ch1011 Module 1. College of the North Atlantic Qatar, n.d. Web. 30 May 2014.
Shrestha, Binod. "CHEM-GUIDE." : Dynamic Nature of Chemical Equilibrium and Its Characteristics. Blogger, n.d. Web. 29 May 2014.
Floden, Mark. "Ch1011 Module 1." Ch1011 Module 1. College of the North Atlantic Qatar, n.d. Web. 30 May 2014.
Shrestha, Binod. "CHEM-GUIDE." : Dynamic Nature of Chemical Equilibrium and Its Characteristics. Blogger, n.d. Web. 29 May 2014.
